GI-Cell opens the way for autoimmune disease treatments by obtaining patent for regulatory T cell large-scale culture technology Patent to enhance GI-Cell’s competitiveness in autoimmune disease treatments
공개 2021-11-30 08:14:15
이 기사는 2021년 11월 30일 08:06 thebell 에 표출된 기사입니다.
GI-Cell, a South Korea-based company specializing in immune cell therapy, announced on November 29 that it obtained a domestic patent for a large-scale culture technology of regulatory T cells for the treatment of autoimmune diseases. With this patent, GI-Cell is expected to become a leader in the autoimmune disease space in the country.Regulatory T cells maintain immune system homeostasis through immunosuppression regulation. Regulatory T cells have been in the spotlight as a therapeutic agent for diseases caused by imbalance of the immune system, such as autoimmune diseases. However, the proportion of regulatory T cells among the total T cells is only about 2-3%, making it difficult to separate and culture cells. It is also difficult to develop therapeutic agents because the characteristic properties of regulatory T cells are easily lost during the culturing process. However, GI-Cell succeeded in maintaining highly functional regulatory T cells through its unique and differentiated culture technology with improved productivity.
FoxP3, a key marker that can confirm the function of regulatory T cells, needs to be continuously expressed during cell culture. GI-Cell confirmed that FoxP3 expression was maintained in almost all cells even after the culture was finished, and the regulatory T cells were successfully cultured up to tens of billions of cells in the research stage. In addition, the regulatory T cells under development at GI-Cell have regulated surface receptor expression to target specific organs or inflammatory lesions (Code name GIC302, Brand name Drone Treg), and can more effectively suppress the inflammatory response within the lesion.
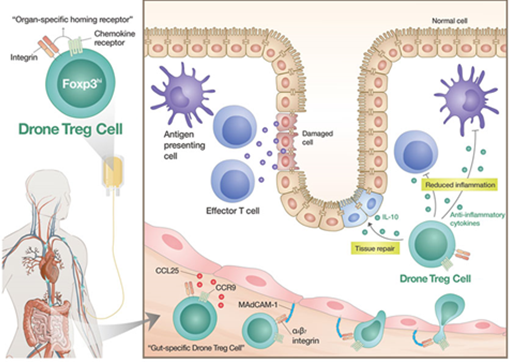
“This patent is recognized for overcoming the limitations of existing regulatory T cell production technology by manufacturing a large amounts of highly active regulatory T cells through a proprietary culture ancillary material and manufacturing methods without changing any characteristic properties of the cells,” said the inventors of the patent, GI Group Chairman Dr. Myoung Ho Jang and GI-Cell CEO Dr. Chun Pyo Hong. “By utilizing Drone Treg platform technology, GI-Cell will spur the development of cell therapies for various autoimmune diseases.”
GI-Cell was recognized in March as an excellent company in job invention compensation by the Korean Intellectual Property Office. This enabled the company to apply for an expedited examination process and register the patent quickly.

< 저작권자 ⓒ 자본시장 미디어 'thebell', 무단 전재, 재배포 및 AI학습 이용 금지 >
best clicks
최신뉴스 in 전체기사
-
- '재무개선' AJ네트웍스, 조달비용 '확' 낮췄다
- '9년만에 엑시트' 한앤코, 한온시스템 거래구조 살펴보니
- 한국타이어앤테크놀로지, 한온시스템 인수한다
- [수술대 오른 커넥트웨이브]2대주주 지분매입 나선 MBK, 주식교환 카드 꺼냈다
- [이사회 모니터]이재용 에이비프로바이오 대표, 바이오·반도체 신사업 '드라이브'
- 와이투솔루션, 주인 바뀌어도 '신약' 중심엔 美 합작사 '룩사'
- 아이티센, 부산디지털자산거래소 본격 출범
- 아이에스시, AI·데이터센터 수주 증가에 '날개'
- [이사회 모니터]서정학 IBK증권 대표, ESG위원회도 참여 '영향력 확대'
- SW클라우드 '10주년' 폴라리스오피스, “초격차 밸류업”




















